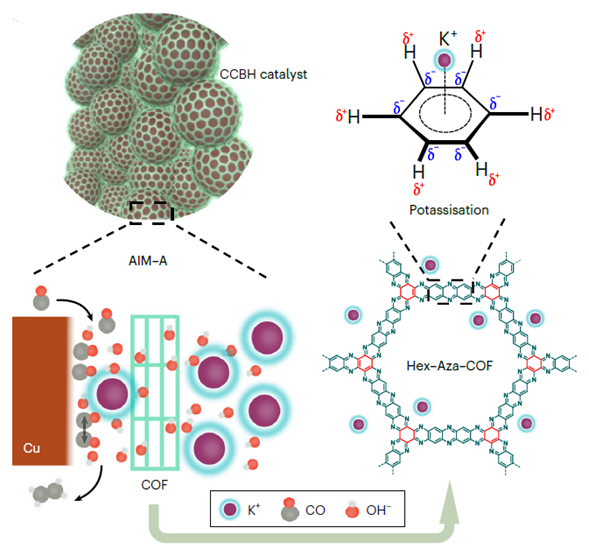
Carbon dioxide/monoxide (CO2/CO) electrolysis provides a pathway to convert CO2 into multicarbon products. However, the energy and carbon efficiencies are low due to the uncontrolled gas/ion distributions in the local reaction environment. In the best current zero-gap CO2RR/CORR electrolyzers, cations (K+) crossover from the anode to the cathode, accumulate on the catalyst surface, block the active sites, limit CO adsorption and lead to hydrogen evolution reaction. This is the primary cause of low energy efficiency (EE) and single pass carbon efficiency (SPCE). Additionally, OH– migration from the cathode to the anode reduces alkalinity on the cathode therefore diminishing C-C coupling-a key step along the pathway to C2+ products.
Ozden et al. (https://doi.org/10.1038/s41560-022-01188-2) reported a way to block cation migration to the catalyst surface by using a catalyst/covalent organic framework (COF) bulk heterojunction (CCBH). The π-conjugated hydrophobic COFs constrain cation (potassium) diffusion via cation–π interactions, while promoting anion (hydroxide) and gaseous feedstock adsorption on the catalyst surface. The CORR performance was evaluated using a commercial CO2RR MEA electrolyzer (Dioxide Materials) and 1M KOH as the anolyte. Bare Cu catalyst showed a peak SPCEc2+ at am EEc2+ of 25% and C2+ current density of 137mA/cm2, while CCBH catalyst achieved a SPECc2+ of 95% at an EEc2+ of 41% and C2+ current density of 210mA/cm2 (Figure 1a). Most importantly, the cell performance was stable for 200hr at a current density of 200mA/cm2 (Figure 1c). In addition, the CCBH catalysts demonstrated superior performance as compared to bare Cu catalyst in neutral CO2RR with Sustainion® anion exchange membrane and acidic CO2RR with Nafion membrane (Figure 1d, e).
Want to get reliable performance of your novel catalysts, try Dioxide Materials Sustainion® membrane and electrolyzer hardware.
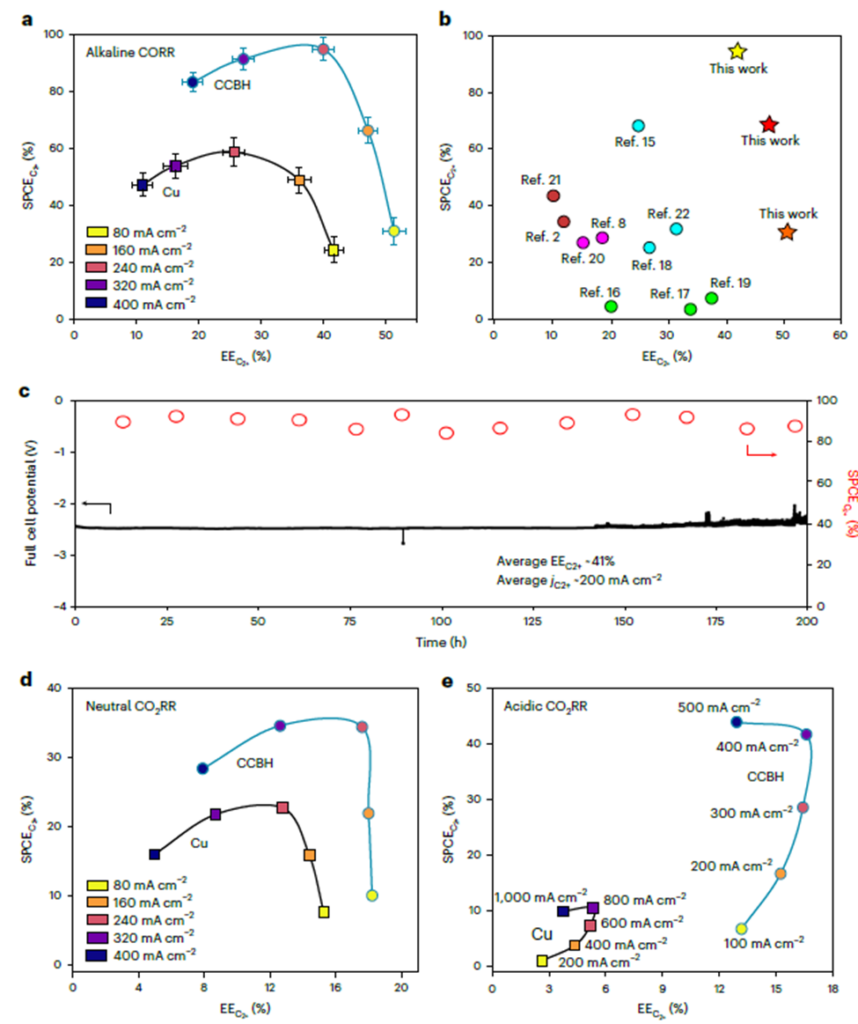
Figure 1 The CCBH catalyst for energy- and carbon-efficient CO2RR/CORR